Trisulfide Reaction: Chemistry Breakthrough
Trisulphide Metathesis Reaction Opens New Chemical Possibilities
Why in the News ?
Scientists from Flinders University, Australia, have discovered a new trisulphide metathesis reaction that allows sulphur–sulphur bonds to break and reform spontaneously at room temperature, offering promising applications in drug modification, recyclable plastics, and advanced chemical manufacturing.
Discovery of the Trisulphide Metathesis Reaction:
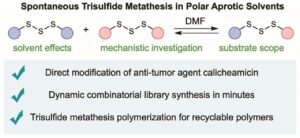
- Researchers recently reported in Nature Chemistry a novel chemical process known as trisulphide metathesis, where molecules containing three sulphur atoms exchange parts with each other.
- These molecules, called organic trisulphides, consist of chains of three sulphur atoms linked together.
- During the reaction, the sulphur–sulphur bonds break and reform rapidly, allowing molecules to swap components with remarkable precision.
- Unlike most chemical reactions involving sulphur bonds, this process occurs spontaneously at room temperature, without the need for extreme heat, light, or catalysts.
- The reaction requires only a common industrial solvent such as dimethylformamide (DMF) and can occur within seconds, making it efficient and practical for laboratory and industrial use.
Potential Applications in Medicine and Materials
- One key application demonstrated by researchers was the modification of the anti-cancer drug calicheamicin, a fragile molecule with a complex chemical structure.
- The new reaction allowed scientists to alter the drug without damaging its delicate molecular framework, which is difficult using conventional chemical techniques.
- Researchers also used the reaction to create a new type of plastic polymer.
- Because the sulphur bonds can be easily broken and reformed, the plastic material can be fully chemically recycled, enabling manufacturers to convert it back into its original raw materials.
- Such innovations could support sustainable materials science, helping reduce plastic waste and promote circular manufacturing systems.
About Metathesis Reactions in Chemistry:● A metathesis reaction refers to a chemical process where two molecules exchange parts to form new compounds. ● Metathesis reactions are widely used in organic chemistry, polymer production, and pharmaceutical research. ● Traditional sulphur chemistry often involves highly reactive radicals or charged ions, which can produce unwanted by-products. ● In this newly discovered process, the reaction occurs through a temporary molecular structure called thiosulphoxide, enabling precise and clean bond exchange. ● The discovery highlights the importance of innovative reaction pathways in advancing green chemistry, recyclable materials, and modern drug development. |